


For most genetic diseases CRISPR is still limited and unable to make the required edits. For example, to treat dominant indications, one allele needs to be edited while leaving the other one intact, and current CRISPR nucleases are not specific enough to do this. Since dominant indications are the vast majority of genetic diseases, a novel approach is required—a next-generation CRISPR.
At EmendoBio, we’ve designed a novel approach that enables us to target any gene. We recognized that a panel of novel nucleases with various characteristics could unlock broader areas of the genome, and that these nucleases can then be optimized per target sequence to achieve the highest levels of precision.
EmendoBio’s OMNI™ technology platform enables extreme precision gene editing customized to any target
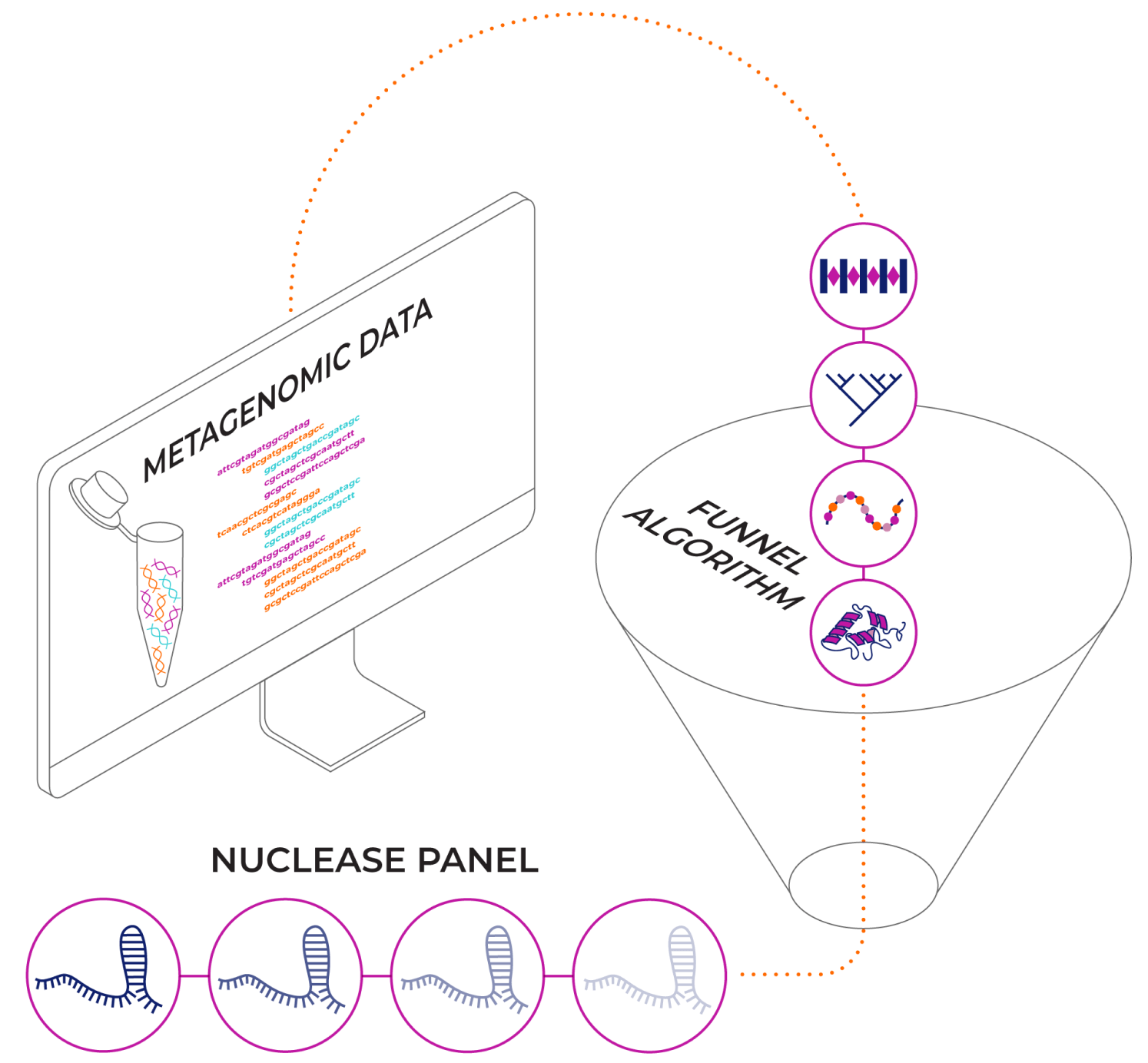

Novel CRISPR Nuclease Discovery
Our cutting-edge computational team scans the vast space of bacteria to identify unknown nucleases. Then, our nuclease discovery team singles out the ones with robust, high activity in mammalian cells.
- Novel OMNI™ nucleases have various desired properties.
- OMNI nucleases are adaptable to a variety of in-vivo delivery systems as well as ex-vivo cell therapy.
- The diversity of OMNI nucleases, particularly novelty of PAM sites, widens our genomic accessibility – Making Any Gene Targetable.
- Non-NGG PAM nucleases enable us to target genomic sites that are not accessible by common spCas9 nucleases.


Nuclease Optimization
The discovery of a panel of unique and rare OMNI nucleases is followed by a process of nuclease optimization. CRISPR gene editing requires the highest efficiency and precision to eliminate off-target effects and enable allele specific editing. Available CRISPR tools can seldom achieve this; in order to achieve allele specificity while maintaining high activity and zero off-target effects, optimization is necessary.
Our computational and nuclease characterization teams create libraries of nuclease variants using a combination of computational AI based predictions and rational design, followed by selection for optimal activity, specificity and stability using specialized assays.
Our OMNI technology platforms result in nucleases optimized per target gene to meet all requirements for successful high precision gene-editing: highly active, allele specific nucleases with no detectable off targets, presenting an exceptional highest precision editing for each target of interest.


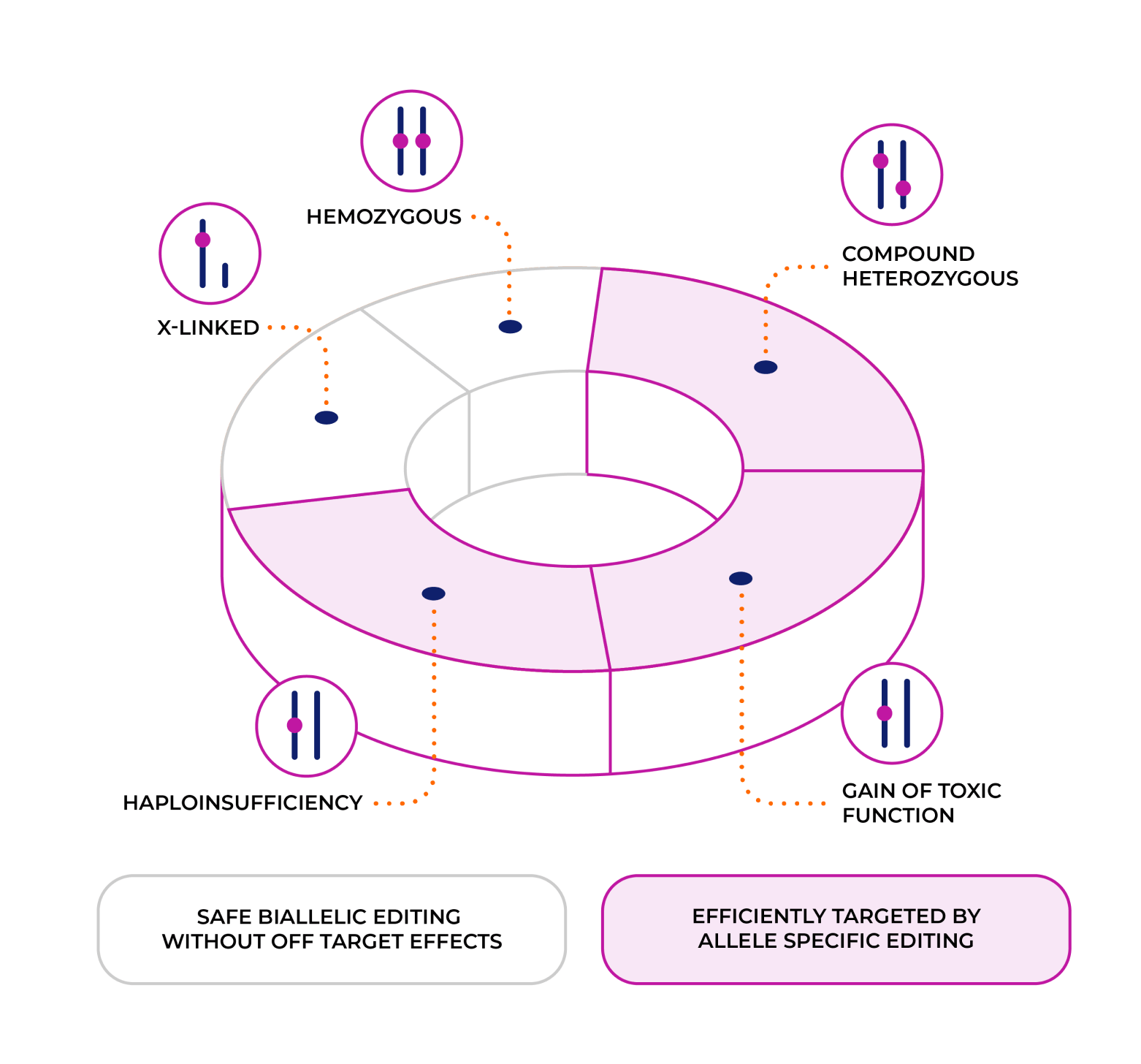

Benefits of EmendoBio’s Extreme Precision Editing
In addition to enabling safe and effective biallelic cleavage, this high precision enables EmendoBio to perform allele specific editing and employ novel gene editing strategies to create commercially viable solutions for genes with numerous mutations.
As a result, EmendoBio is uniquely positioned to treat indications that are dominant, dominant negative, and compound heterozygous, covering the majority of genetic disorders that other technologies cannot address.
The fully in-house developed, state-of-the-art platforms employed by EmendoBio have brought CRISPR technology to the next level. We can now take on challenging diseases that are not addressable with first generation CRISPR technologies. We are able to design tailor-made nucleases and RNA guides for many diseases that were previously considered incurable.
Lead Indication – Allele Specific Editing to Cure Dependent Severe Congenital Neutropenia (SCN)
- Curative approach for highly unmet need
- Program performing allele specific editing, a capability demonstrated uniquely by EmendoBio
- An engineered variant eliminating off-target and showing complete allele specificity
Neutrophil Maturation Disorder
- Dominant mutations in ELANE gene cause protein misfolding, ER stress and maturation arrest
- Prevalence 1:250,000 but likely under-diagnosed
- ~16,000 patients worldwide
- Severe recurrent infections
High Unmet Need
- Lifelong daily injection of G-CSF has severe side effects, increases the risk for AML/MDS in a dose dependent manner, not curative.
- Allo-transplants are associated with 17% transplant related mortality, 10% graft failure and 20% acute GvhD
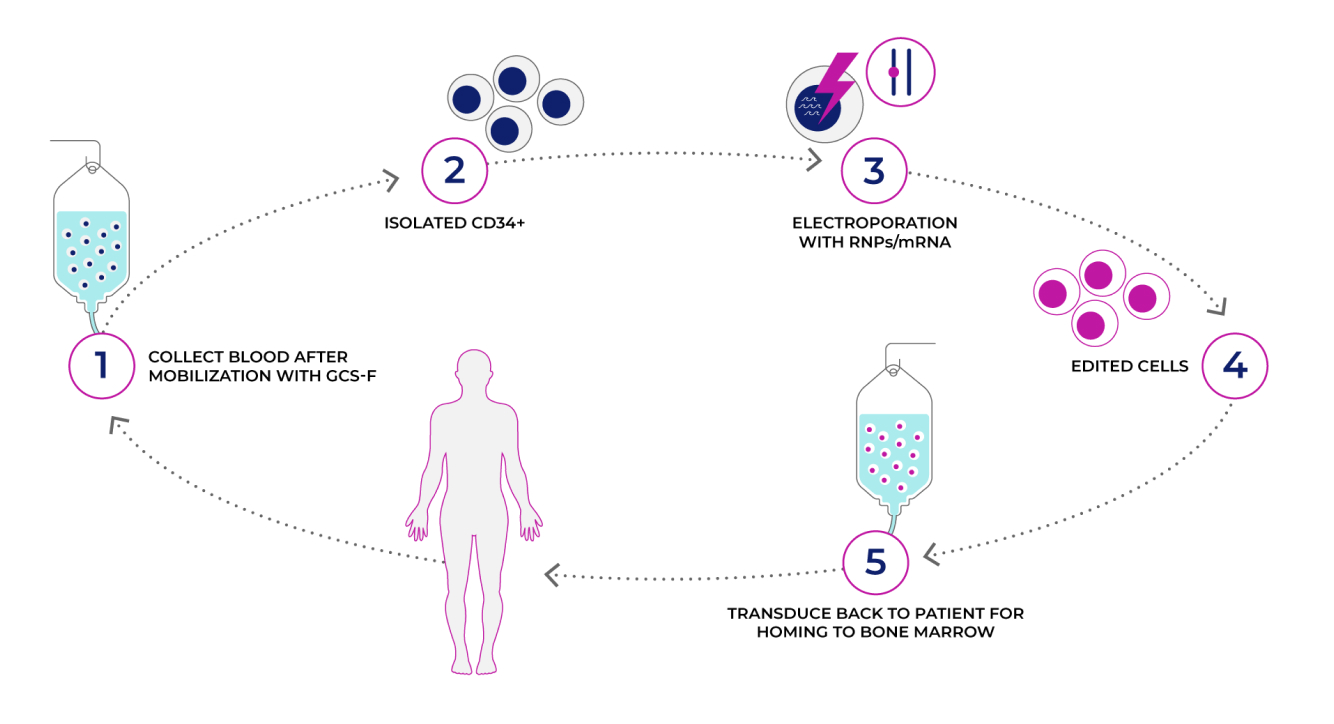
EmendoBio’s Treatment Strategy
- Autologous HSPCs product
- Ex-vivo editing
- Allele-specific Cas nuclease
- Two sgRNA
- Single nucleotide polymorphism (SNP) based editing strategy
- ELANE expressed exclusively in neutrophils
- Survival advantage – low level of correction is expected to be sufficient
- Clear and short-term clinical end points
- Curative
- Patient-derived HSCs were edited ex-vivo only on the targeted mutated allele and differentiated into functional neutrophils to achieve therapeutic effect.